Research Archive
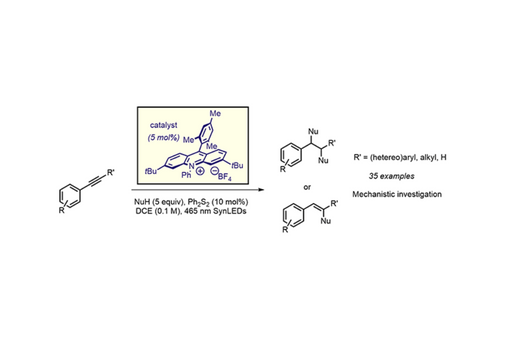
Herein, we communicate a strategy for the divergent functionalization of alkynes with photoexcited acridinium organic dyes, presumably via the formation of vinyl cation radicals as key intermediates.
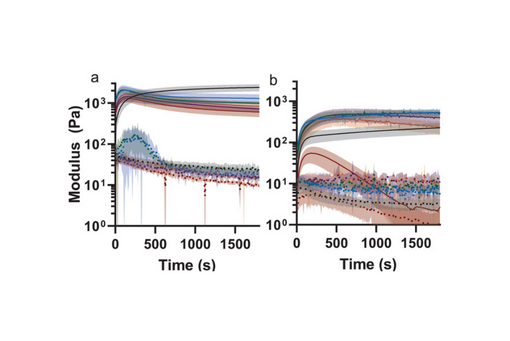
A series of injectable polysaccharide hydrogels are prepared with oxidized dextran and diethylenetriamine-modified carboxymethylcellulose (CMC) or hyaluronic acid (HA).
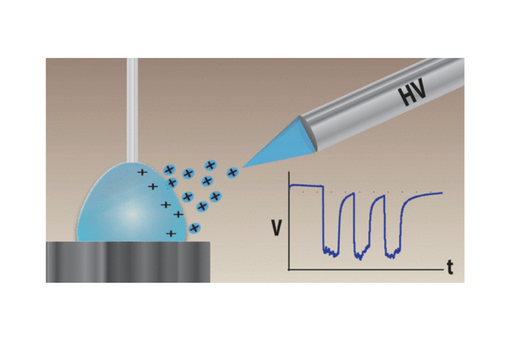
The current work introduces a new method utilizing PILSNER for the examination of the particles generated during ESI using simple analysis techniques with a commercially available potentiostat.
To this end we introduce analysis of liquid particles in aerosols via charge-induction amperometry (ALPACA), an extremely simple potentiostat-based technique capable of on-line, rapid measurement of the aggregate charge of aerosol particles.
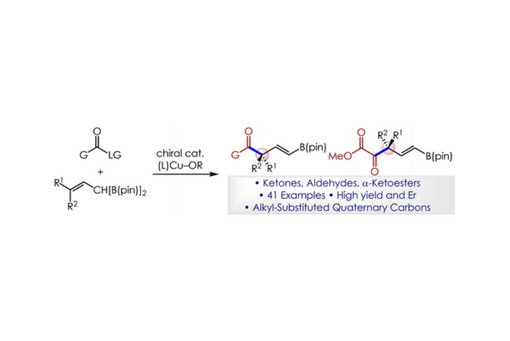
A practical method for the efficient and enantioselective preparation of versatile ketones and aldehydes that contain an α-quaternary stereocenter is described.
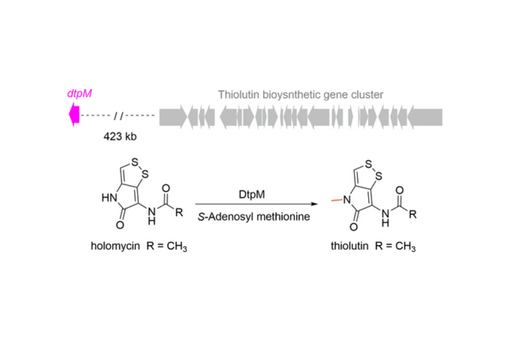
Here, we identified and characterized the amide N-methyltransferase DtpM that is encoded >400 kb outside of the thiolutin gene cluster.
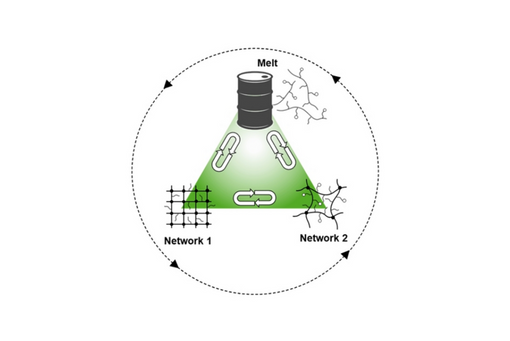
The crosslink density was tuned by loading the functionalized networks with a controlled fraction of dormant crosslinkers and crosslinker scavengers, such as furan-capped bis-maleimide and anthracene, respectively.
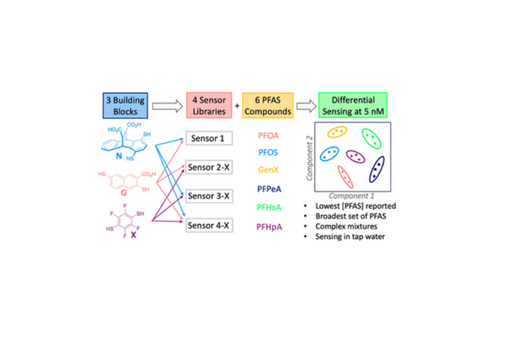
Here we show that dynamic combinatorial libraries (DCLs) with simple building blocks can be templated with a fluorophore and subsequently used as sensors to form an array that differentially detects each PFAS species and various mixtures thereof.
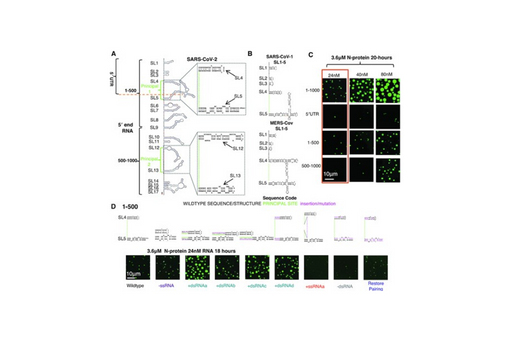
We identify two separate and distinct double-stranded, RNA motifs (dsRNA stickers) that promote N-protein condensation.
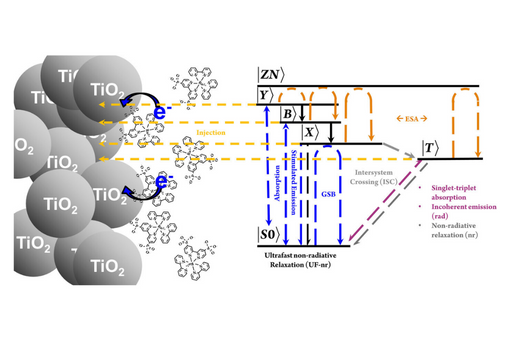
A kinetic framework for the ultrafast photophysics of tris(2,2-bipyridine)ruthenium(II) phosphonated and methyl-phosphonated derivatives is used as a basis for modeling charge injection by ruthenium dyes into a semiconductor substrate.
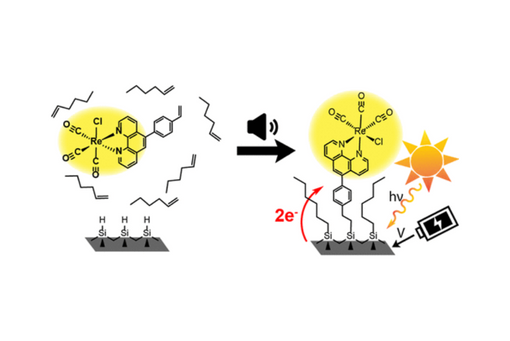
A sonochemical-based hydrosilylation method was employed to covalently attach a rhenium tricarbonyl phenanthroline complex to silicon(111).
First-principles dynamics simulation is used to study how excited electron dynamics in a type-II semiconductor–molecule interface is affected when the surface of a bulk semiconductor is replaced with that of a quantum dot.

