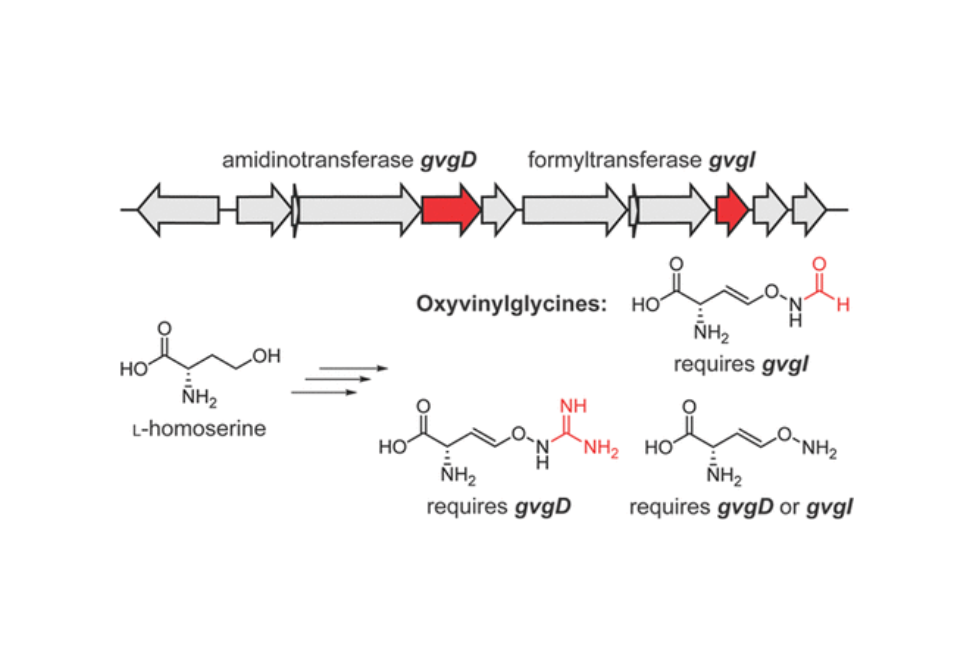
Research Archive
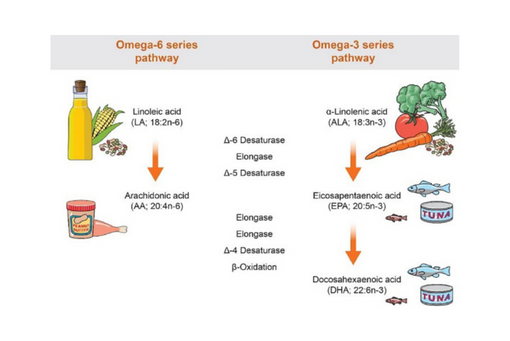
This cross-sectional study examined relationships between PUFAs in circulating erythrocytes and 2 chronic idiopathic pain conditions: temporomandibular disorder (TMD) and low back pain in a community-based sample of 503 U.S. adults.
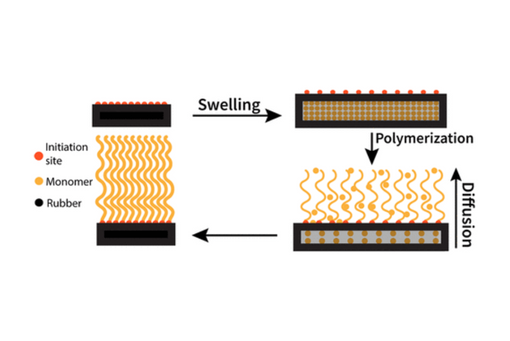
Here we use a combination of coarse-grained computer simulations and experimental techniques to demonstrate how this approach can be implemented, with swellable network-like substrates supplying monomers to the functionalized surface as they diffuse out of the swollen network.
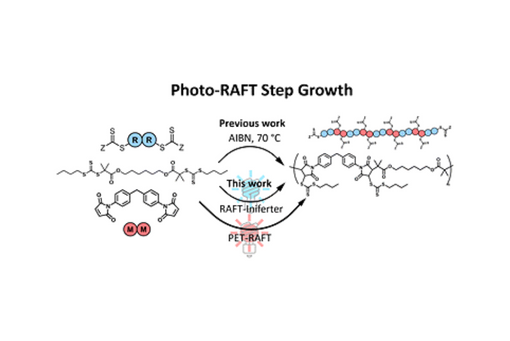
Photomediated RAFT step-growth polymerization was performed with and without the presence of a photocatalyst using a trithiocarbonate-based CTA and a maleimide monomer.
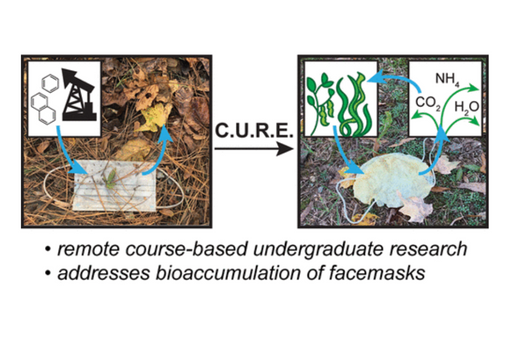
Here, through an at-home course-based undergraduate research experience (CURE), we demonstrate that alginate and soy-based materials are attractive alternatives for mask fabrication: stacking calcium ion-cross-linked alginate films with soy protein isolate sheets enables electrostatic and size-exclusion filtration.
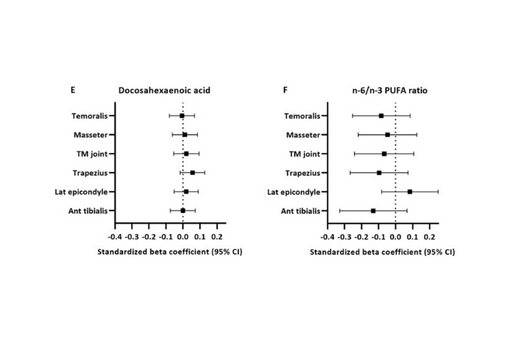
This study sought to determine whether free PUFAs found in red blood cells also play a role in nociceptive processing.
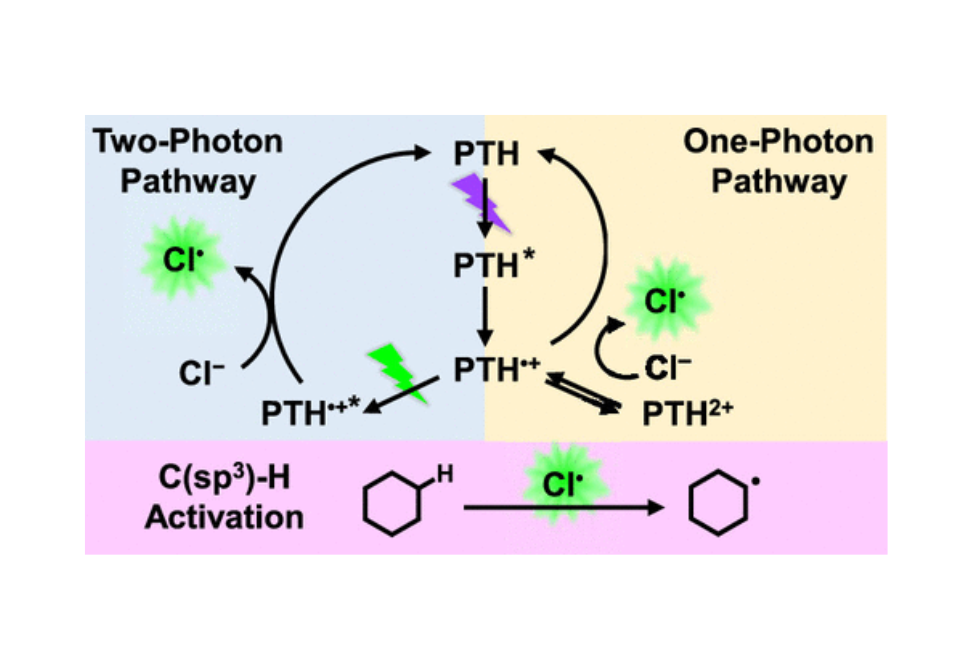
Herein, two photocatalytic chloride oxidation pathways that involve either one- or consecutive two-photon excitation of N-phenylphenothiazine (PTH) are presented.
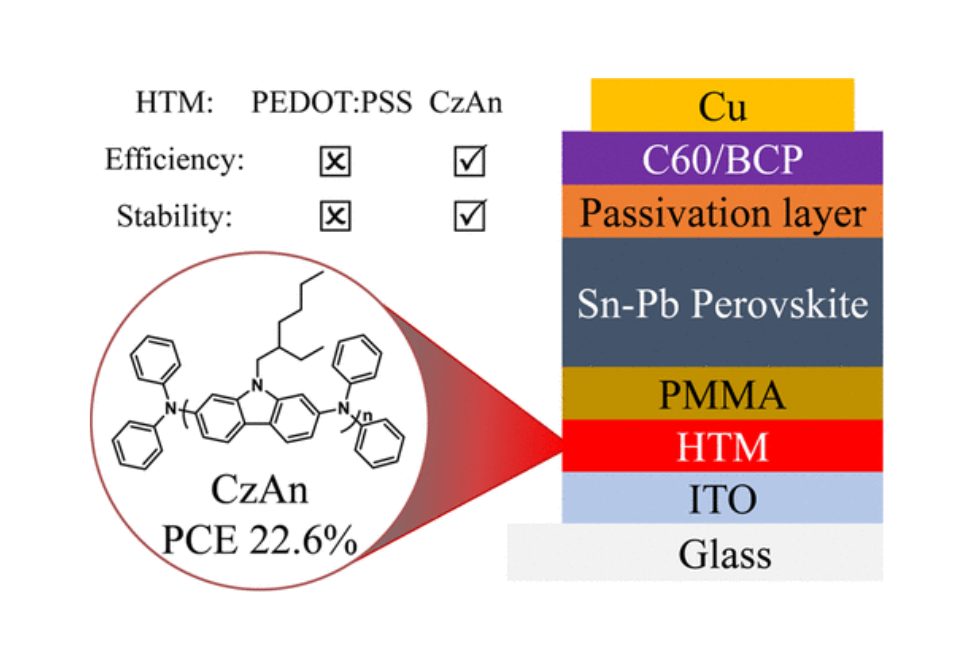
Herein, an easily synthesized polymer HTM poly[(phenyl)imino[9-(2-ethylhexyl)carbazole]-2,7-diyl] (CzAn) with a shallow highest occupied molecular orbital (HOMO) level of −4.95 eV is used in a p-i-n structure, methylammonium-free, Sn–Pb PSC to replace PEDOT:PSS.