Selecting the appropriate indirect viability assay for 3D paper-based cultures: a data-driven study
Abstract
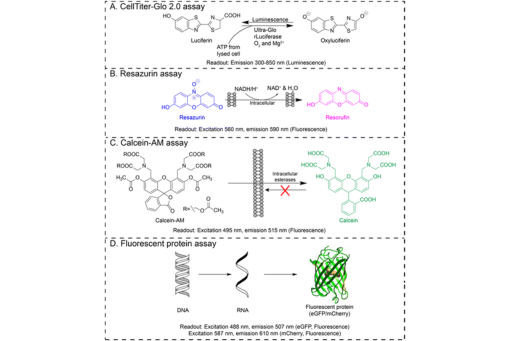
Cellular viability measurements quantify decreased proliferation or increased cytotoxicity caused by drug candidates or potential environmental toxins. Direct viability measures count each cell to provide an accurate readout. This approach can prove analytically challenging and time-consuming when cells are maintained in 3D structures akin to tissues or solid tumors. While less labor-intensive, indirect viability measures can be less accurate due to the heterogeneous structural and chemical microenvironment that arises when cells are maintained in tissue-like architectures and in contact with extracellular matrices. Here we determine the analytical figures of merit of five indirect viability assays in the paper-based cell culture platform we continue to develop in our laboratory: calcein-AM staining, the CellTiter-Glo assay, imaging fluorescent protein expression, propidium iodide staining, and the resazurin assay. We also determined the compatibility of each indirect assay with hypoxic conditions, intra-experimental repeatability, inter-experimental reproducibility, and ability to predict a potency value for a known antineoplastic drug. Our results show that each assay has benefits and drawbacks to consider when choosing the appropriate readout to answer a particular research question. We also highlight that only one indirect readout is unaffected by hypoxia, a commonly overlooked variable in cell culture that likely yields inaccurate viability measures.
Citation
Sitte, Z. R., Larson, T. S., McIntosh, J. C., Sinanian, M., & Lockett, M. R. (2023). Selecting the appropriate indirect viability assay for 3D paper-based cultures: A data-driven study. The Analyst, 148(10), 2245–2255. https://doi.org/10.1039/d3an00283g