Mechanistic investigation of a visible light mediated dehalogenation/cyclisation reaction using iron(iii), iridium(iii) and ruthenium(ii) photosensitizers
Abstract
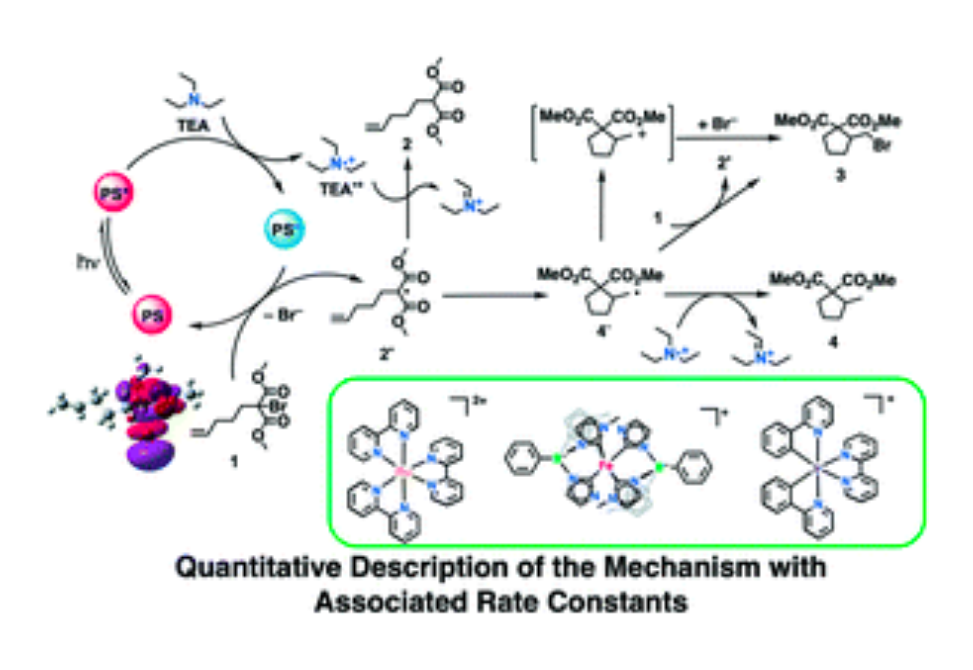
The mechanism of a visible light-driven dehalogenation/cyclization reaction was investigated using ruthenium(ii), iridium(iii) and iron(iii) photosensitizers by means of steady-state photoluminescence, time-resolved infrared spectroscopy, and nanosecond/femtosecond transient absorption spectroscopy. The nature of the photosensitizer was found to influence the product distribution such that the dehalogenated, non-cyclized products were only detected for the iron photosensitizer. Strikingly, with the iron photosensitizer, large catalytic yields required a low dielectric solvent such as dichloromethane, consistent with a previous publication. This low dielectric solvent allowed ultrafast charge-separation to outcompete geminate charge recombination and improved cage escape efficiency. Further, the identification of reaction mechanisms unique to the iron, ruthenium, and iridium photosensitizer represents progress towards the long-sought goal of utilizing earth-abundant, first-row transition metals for emerging energy and environmental applications.
Citation
Mechanistic investigation of a visible light mediated dehalogenation/cyclisation reaction using iron(III), iridium(III) and ruthenium(II) photosensitizers
A. Aydogan, R. E. Bangle, S. De Kreijger, J. C. Dickenson, M. L. Singleton, E. Cauët, A. Cadranel, G. J. Meyer, B. Elias, R. N. Sampaio and L. Troian-Gautier, Catal. Sci. Technol., 2021, 11, 8037 DOI: 10.1039/D1CY01771C