
Albert A Bowers
Adjunct Associate Professor
Marsico 3017(919) 962-4336
abower2@email.unc.edu
Group Website
Research Interests
Discovery of next-generation peptide therapeutics
Research Synopsis
We are a research lab in the Division of Chemical Biology and Medicinal Chemistry (CBMC) at UNC Chapel Hill. Our research focuses on the discovery and development of peptide macrocycles as next-generation therapeutics. We exploit mRNA display, a biochemical method for the rapid synthesis and screening of vast peptide libraries, as our central discovery engine in this work. Research is generally split between methods and targets: 1) developing methods to improve the diversity and pharmacological profiles of macrocycles available to mRNA display and 2) deploying these methods against important therapeutic targets and target classes that currently lack ligands and therapeutic starting points. These efforts involve the integration of medicinal chemistry, biochemistry, molecular biology, bioinformatics, and computational chemistry. More information can be found on the research page. We are also members of the Department of Chemistry, the Center for Integrative Chemical Biology and Drug Discovery, and the Lineberger Comprehensive Cancer Center.
Professional Background
BA, The University of Chicago, 2001; PhD, University of Illinois at Chicago, 2007; NIH Postdoctoral Fellow, Harvard Medical School, 2008-2011; Beckman Young Investigator Award, 2014-2018; Boulder Peptide Society Young Investigator Award, 2016.
News & Publications
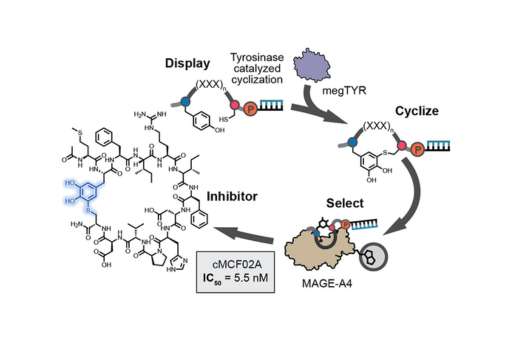
Here we show that peptides containing tyrosine and cysteine are rapidly cyclized upon tyrosinase treatment.
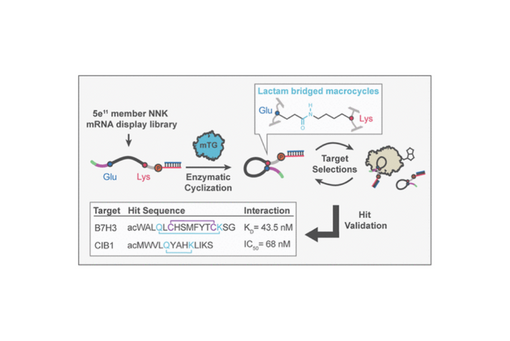
Here, we report the evaluation and implementation of the common industrial enzyme, microbial transglutaminase (mTG), as a versatile biocatalyst for cyclization of mRNA display peptide libraries via lysine-to-glutamine isopeptide bonds.

