Rapid, inexpensive, sequence-independent fluorescent labeling of phosphorothioate DNA
Abstract
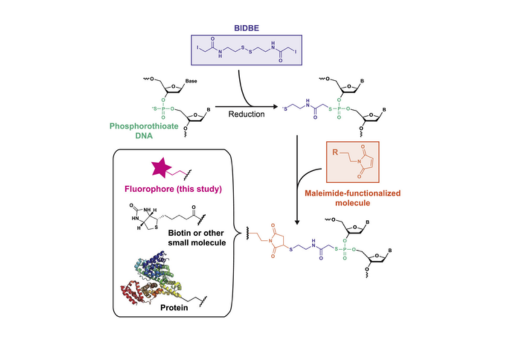
Fluorescently labeled oligonucleotides are powerful tools for characterizing DNA processes; however, their use is limited by the cost and sequence requirements of current labeling technologies. Here, we develop an easy, inexpensive, and sequence-independent method for site-specifically labeling DNA oligonucleotides. We utilize commercially synthesized oligonucleotides containing phosphorothioate diester(s) in which a nonbridging oxygen is replaced with a sulfur (PS-DNA). The increased nucleophilicity of the thiophosphoryl sulfur relative to the phosphoryl oxygen permits selective reactivity with iodoacetamide compounds. As such, we leverage a long-existing bifunctional linker, N,N'-bis(α-iodoacetyl)-2-2'-dithiobis(ethylamine) (BIDBE), that reacts with PS-DNAs to leave a free thiol, allowing conjugation of the wide variety of commercial maleimide-functionalized compounds. We optimized BIDBE synthesis and its attachment to PS-DNA and then fluorescently labeled the BIDBE-PS-DNA using standard protocols for labeling cysteines. We purified the individual epimers, and using single-molecule Förster resonance energy transfer (FRET), we show that the FRET efficiency is independent of the epimeric attachment. Subsequently, we demonstrate that an epimeric mixture of double-labeled Holliday junctions (HJs) can be used to characterize their conformational properties in the absence and presence of the structure-specific endonuclease Drosophila melanogaster Gen. Finally, we use a biochemical activity assay to show that this double-labeled HJ is functional for cleavage by Gen and that the double-labeled HJ allows multiple DNA species to be identified in a single experiment. In conclusion, our results indicate that dye-labeled BIDBE-PS-DNAs are comparable to commercially labeled DNAs at a significantly reduced cost. Notably, this technology could be applied to other maleimide-functionalized compounds, such as spin labels, biotin, and proteins. The sequence independence of labeling, coupled with its ease and low cost, enables unrestricted exploration of dye placement and choice, providing the potential for creation of differentially labeled DNA libraries and opening previously inaccessible experimental avenues.
Citation
Satusky, M. J., Johnson, C. V., & Erie, D. A. (2023). Rapid, inexpensive, sequence-independent fluorescent labeling of phosphorothioate DNA. Biophysical Journal, 122(7), 1211–1218. https://doi.org/10.1016/j.bpj.2023.02.011