Mechanistic Investigations into Amination of Unactivated Arenes via Cation Radical Accelerated Nucleophilic Aromatic Substitution
Abstract
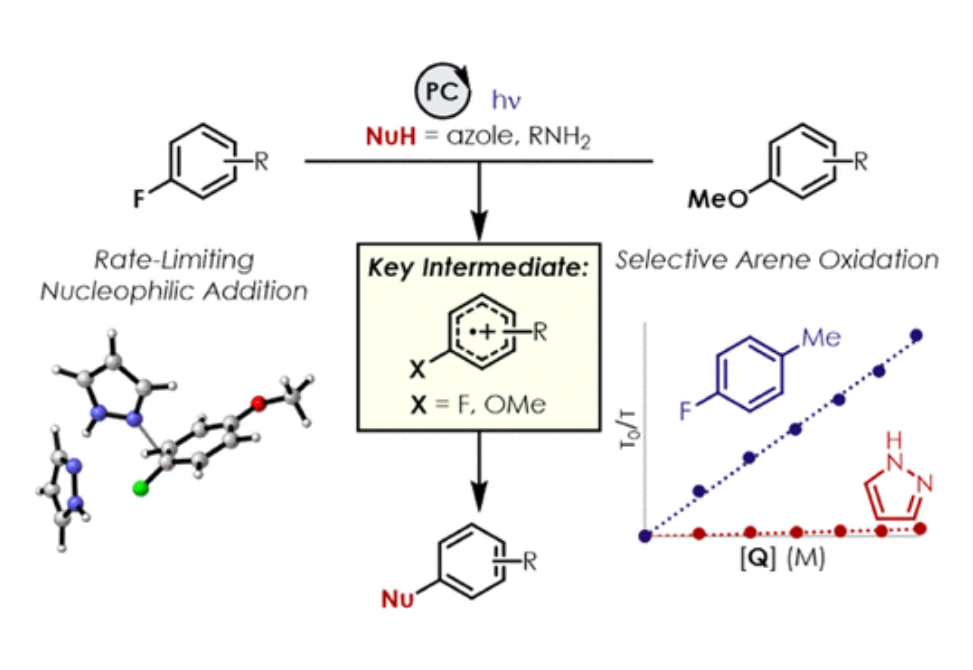
A mechanistic investigation into the amination of electron-neutral and electron-rich arenes using organic photoredox catalysis is presented. Kinetic and computational data support rate-limiting nucleophilic addition into an arene cation radical using both azole and primary amine nucleophiles. This finding is consistent with both fluoride and alkoxide nucleofuges, supporting a unified mechanistic picture using cation radical accelerated nucleophilic aromatic substitution (CRA-SNAr). Electrochemistry and time-resolved fluorescence spectroscopy confirm the key role solvents play in enabling selective arene oxidation in the presence of amines. The synthetic limitations of xanthylium salts are elucidated via photophysical studies. An alternative catalyst scaffold with improved turnover numbers is presented.
Citation
Mechanistic Investigations into Amination of Unactivated Arenes via Cation Radical Accelerated Nucleophilic Aromatic Substitution
Vincent A. Pistritto, Shubin Liu, and David A. Nicewicz
Journal of the American Chemical Society 2022 144 (33), 15118-15131
DOI: 10.1021/jacs.2c04577