Mapping Solvent Entrapment in Multiphase Systems by Electrogenerated Chemiluminescence
Abstract
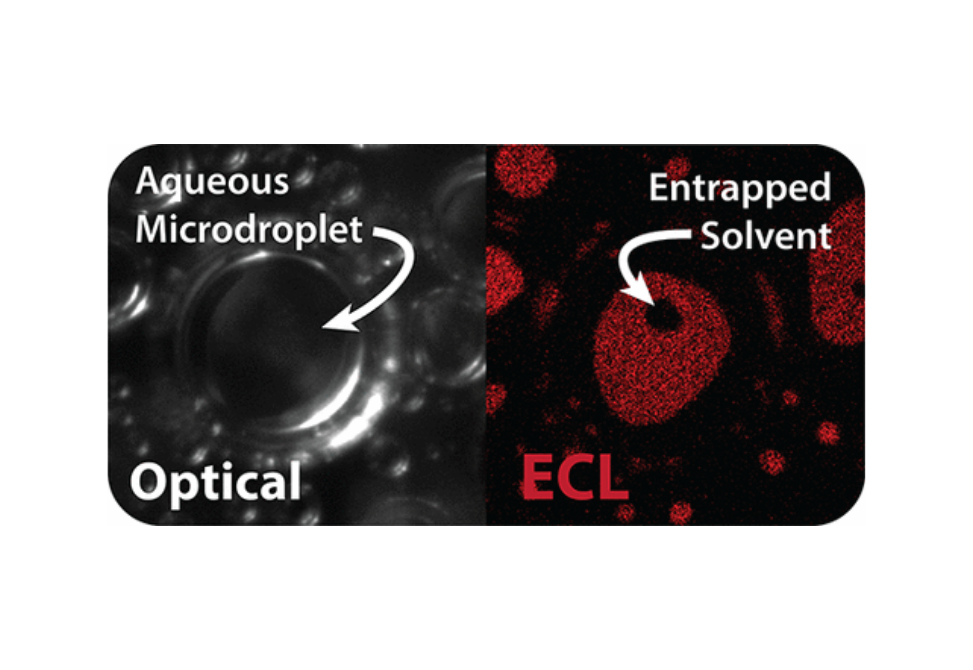
The interfacial properties of multiphase systems are often difficult to quantify. We describe the observation and quantification of immiscible solvent entrapment on a carbonaceous electrode surface using microscopy-coupled electrogenerated chemiluminescence (ECL). As aqueous microdroplets suspended in 1,2-dichloroethane collide with a glassy carbon electrode surface, small volumes of the solvent become entrapped between the electrode and aqueous phase, resulting in an overestimation of the true microdroplet/electrode contact area. To quantify the contribution of solvent entrapment decreasing the microdroplet contact area, we drive an ECL reaction within the microdroplet phase using tris(bipyridine)ruthenium(II) chloride ([Ru(bpy)3]Cl2) as the ECL luminophore and sodium oxalate (Na2C2O4) as the co-reactant. Importantly, the hydrophilicity of sodium oxalate ensures that the reaction proceeds in the aqueous phase, permitting a clear contrast between the aqueous and 1,2-dichloroethane present at the electrode interface. With the contrast provided by ECL imaging, we quantify the microdroplet radius, apparent microdroplet contact area (aqueous + entrapped 1,2-dichloroethane), entrapped solvent contact area, and the number of entrapped solvent pockets per droplet. These data permit the extraction of the true microdroplet/electrode contact area for a given droplet, as well as a statistical assessment regarding the probability of solvent entrapment based on microdroplet size.
Citation
Mapping Solvent Entrapment in Multiphase Systems by Electrogenerated Chemiluminescence
Matthew W. Glasscott, Silvia Voci, Philip J. Kauffmann, Andrei I. Chapoval, and Jeffrey E. Dick
Langmuir 2021 37 (9), 2907-2912
DOI: 10.1021/acs.langmuir.0c03445