Leakless, Bipolar Reference Electrodes: Fabrication, Performance, and Miniaturization
Abstract
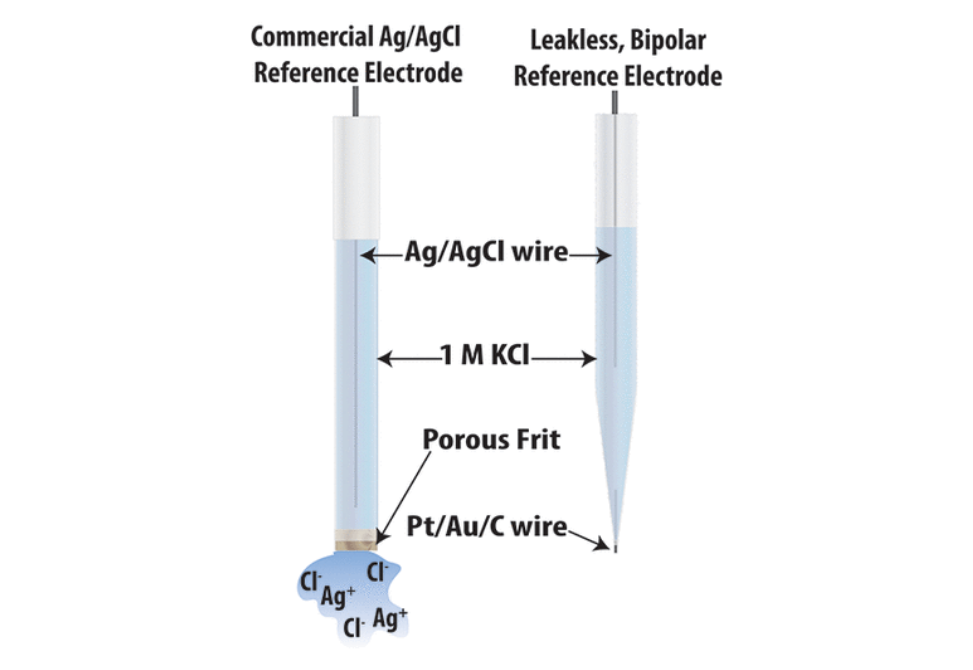
Reference electrodes must maintain a well-defined potential for long periods of time to be useful. The silver/silver chloride (Ag/AgCl) reference electrode is arguably the most widely used reference electrode, but it leaks silver and chloride ions into the sample solution through the porous frit over time. Further, the porous frit makes miniaturization to the micro- and nanoscale challenging. Here, we present an alternative, where the traditional Ag/AgCl reference electrode porous frit is replaced by a conductive wire, preventing ion leakage and allowing miniaturization to the microscale. Charge balance is maintained through a closed bipolar electrochemical mechanism, where faradaic processes occur on each end of the sealed wire. Using the above design, we demonstrate the efficacy of the leakless, bipolar reference electrode (BPRE) and miniaturize it to the microscale (μ-leakless BPRE). Importantly, we demonstrate that leakless and μ-leakless BPREs behave the same as commercial reference electrodes during potentiometric measurements and leakless BPREs perform similarly during voltammetric measurements on ultramicroelectrodes. We demonstrate that the drift during voltammetry using a leakless BPRE on a macroelectrode is slightly more appreciable compared to the drift seen with a commercial reference electrode. We detail design principles for the use of leakless BPREs in nonaqueous solvents and in sealing other conductive materials (e.g., gold and carbon). Using mass spectrometry, we show that the maximum leakage of methylene blue is 0.36 fmol/s, at least 2 orders of magnitude smaller than that of commercial reference electrodes. Finally, we demonstrate the efficacy of using leakless BPREs in potentiometric glucose sensing.
Citation
Leakless, Bipolar Reference Electrodes: Fabrication, Performance, and Miniaturization
Nicole L. Walker and Jeffrey E. Dick
Analytical Chemistry 2021 93 (29), 10065-10074
DOI: 10.1021/acs.analchem.1c00675