Electrodeposition of ligand-free copper nanoparticles from aqueous nanodroplets
Abstract
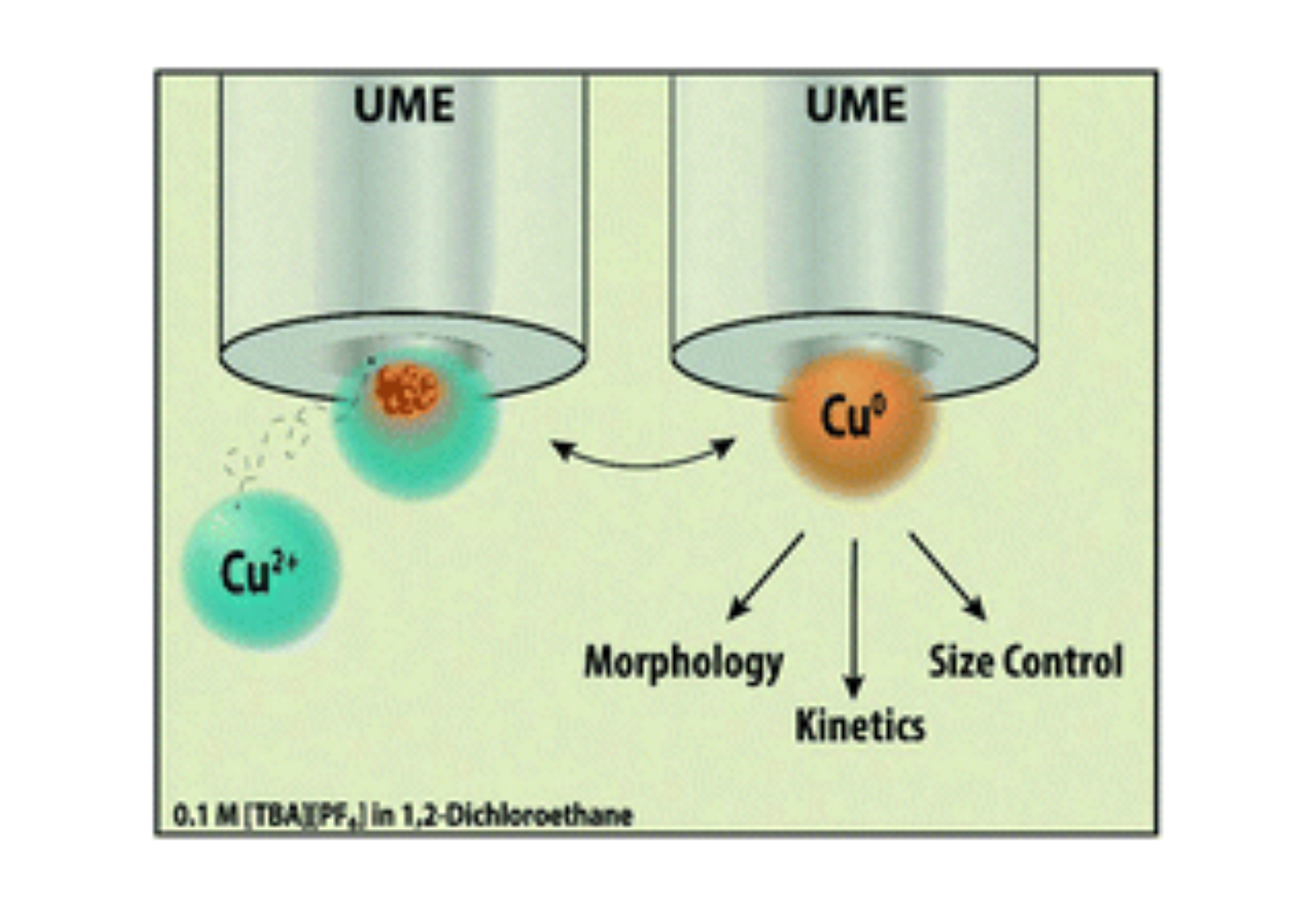
Copper nanoparticles have emerged as promising electrocatalysts for energy storage and conversion. Generally, homogenous nanoparticle synthesis requires a stabilizing ligand, which may influence the electrocatalysis. Ligands can be avoided by direct nanoparticle electrodeposition. Here, we extend nanodroplet-mediated electrodeposition to the electrodeposition of copper nanoparticles from aqueous nanodroplets suspended in 0.1 M tetrabutylammonium hexafluorophosphate ([TBA][PF6]) and 1,2-dichloroethane. Stochastic electrochemistry on microelectrodes was used to elucidate nanoparticle growth kinetics for various solution conditions and substrate materials. A study on the effect of surfactants on nanoparticle morphology and growth kinetics demonstrated the surfactant’s role as an avenue for morphological control. Nanoparticle morphology was studied by Scanning Electron Microscopy (SEM) and Transmission Electron Microscopy (TEM). Energy-dispersive X-ray Spectroscopy (EDX) confirmed the presence of copper nanoparticles. TEM and Selected Area Electron Diffraction (SAED) confirmed nanoparticle size, morphology, and polycrystallinity. We demonstrate tunable nanoparticle size using cyclic voltammetry on single aqueous nanodroplets by altering the voltammetric sweeping potentials of copper electroreduction. Finite element simulations validate the voltammetric response and ability to control the nanoparticle size with nanometer resolution during a voltammetric sweep. These results inform the electrodeposition of copper nanoparticles from aqueous nanodroplets for important applications, such as the conversion of CO2 to hydrocarbon fuels.
Citation
Electrodeposition of ligand-free copper nanoparticles from aqueous nanodroplets