Crystallization-Enabled Henry Reactions: Stereoconvergent Construction of Fully Substituted [N]-Asymmetric Centers
Abstract
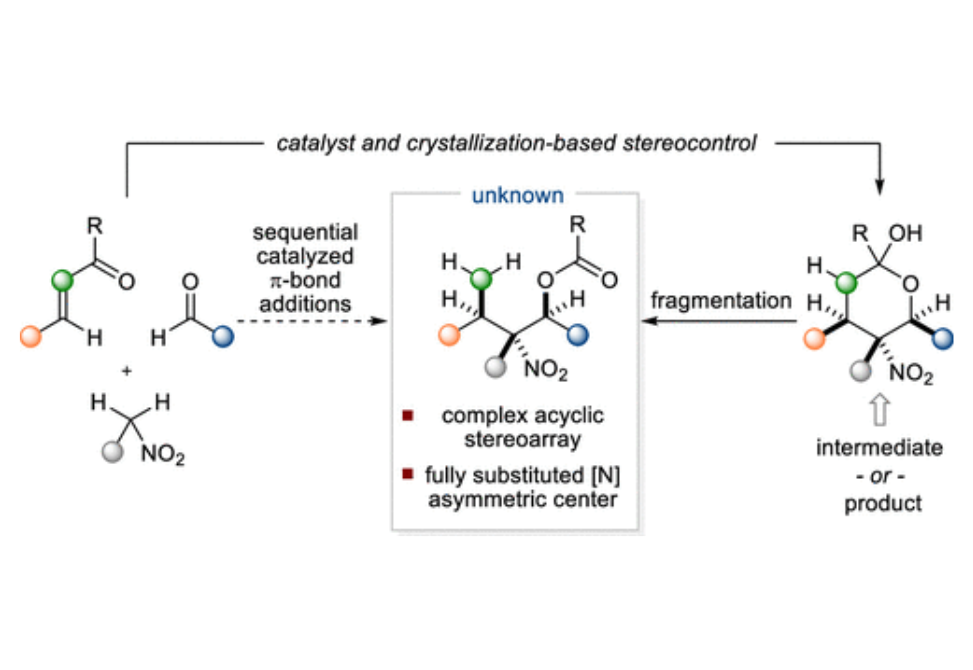
Tetrasubstituted stereogenic carbon centers bearing a nitrogen substituent represent important motifs in medicinal chemistry and natural products; therefore, the development of efficient methods for the stereoselective synthesis of this class of compounds continues to be an important problem. This article describes stereoconvergent Henry reactions of γ,γ-disubstituted nitroalkanes to deliver highly functionalized building blocks containing up to five contiguous stereogenic centers including a fully substituted [N]-asymmetric center. Henry reactions of higher order nitroalkanes are often characterized by their reversibility and minimal accompanying thermodynamic stereocontrol. In contrast, mechanistic studies for the present case suggest a scenario in which reversibility is productively leveraged through crystallization-based stereocontrol, thereby enabling the efficient sequential π-additions of readily accessible starting materials to assemble complex acyclic stereoarrays.
Citation
Crystallization-Enabled Henry Reactions: Stereoconvergent Construction of Fully Substituted [N]-Asymmetric Centers
Pedro De Jesús Cruz and Jeffrey S. Johnson
Journal of the American Chemical Society 2022 144 (34), 15803-15811
DOI: 10.1021/jacs.2c06669