Anodic coulometry of zero-valent iron nanoparticles
Abstract
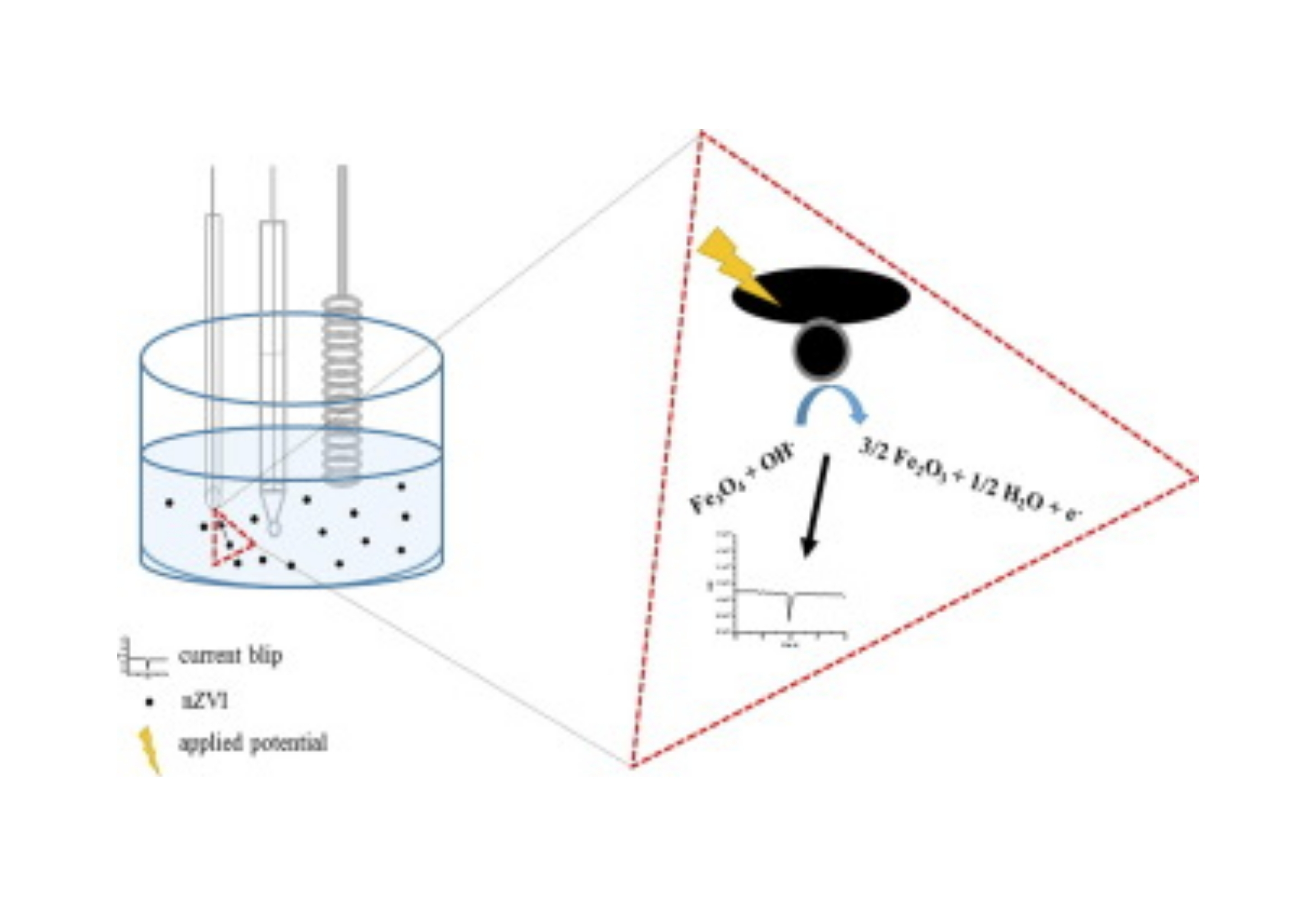
Nanoscale zero-valent iron (nZVI) particles are currently used for environmental remediation due to their ion sequester ability. This trait plus their magnetic behavior, and biocompatibility, makes them a promising alternative for heavy metal poisoning treatment, development of MRI dye, and drug delivery loads, among others. Their rapid passivation in aqueous media inhibit their differentiation from magnetite (Fe3O4) by electrochemical methods, due to its surface oxide. Herein, this work is focused on applying the electrochemical method known as anodic particle coulometry (APC) to detect and characterize nZVI. During the experiments, current blips are observed as a result of a current increase when the electroactive nZVI particle reaches the Au working ultra-microelectrode surface. The integration of current blips resulted in a charge distribution between 4 pC and 5 pC, which correlates with nanoparticles in a size range between 70 nm and 100 nm. The size distribution obtained were compared to the size distribution found using scanning transmission electron microscopy (STEM) and nanoparticle tracking analysis (NTA). The results were compatible with the results obtained by the other two techniques. Characterization of nZVI particles using x-ray photoelectron spectroscopy (XPS) and electron energy loss spectroscopy (EELS) validate the presence of elemental iron which differentiates the material from magnetite. A chemical shift of ca. 1 eV from metallic iron to iron oxide (L3: 708 eV and L2: 721.2 eV versus L3: 709 eV and L2: 722.2 eV) is observed from EELS at nZVI particle center and edge, respectively. This suggests the presence of a passivated surface and a metallic core, a metallic core and oxide shell structure.
Citation
Annelis O. Sánchez-Álvarez, Jeffrey E. Dick, Eduardo Larios, Carlos R. Cabrera,
Anodic coulometry of zero-valent iron nanoparticles,
Journal of Electroanalytical Chemistry,
Volume 896,
2021,
115331,
ISSN 1572-6657,
https://doi.org/10.1016/j.jelechem.2021.115331.