Photocatalytic Transfer Hydrogenation in Water: Insight into Mechanism and Catalyst Speciation
Abstract
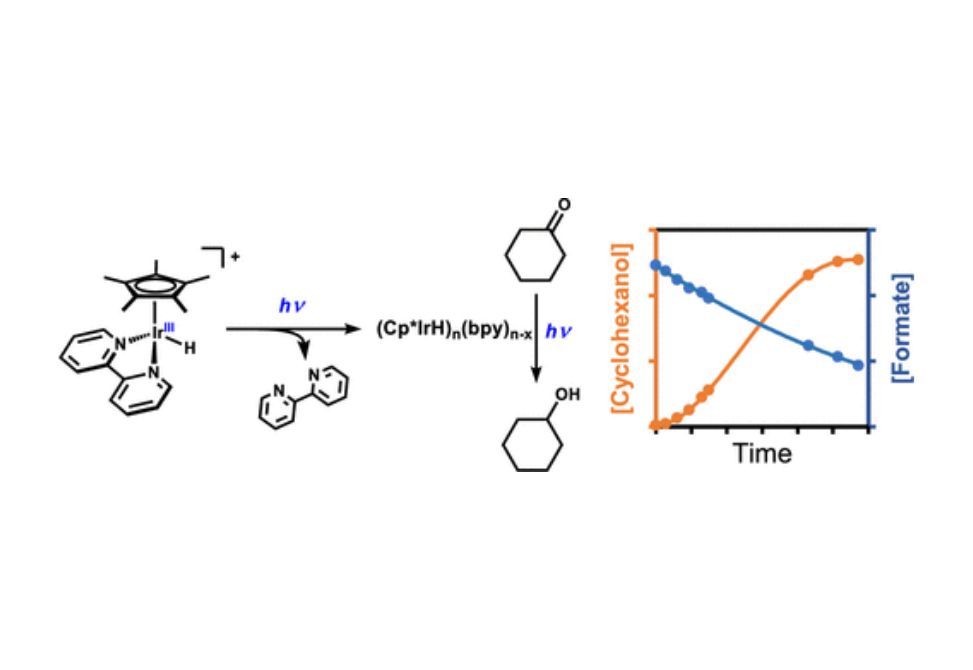
Organometallic catalysts with strong metal-to-ligand charge-transfer (MLCT) optical transitions and reactive metal–hydride bonds provide opportunities for new excited-state reactivity. Toward this end, we have investigated photoinduced transfer hydrogenation reactions for a series of Cp*IrH complexes with functionalized bipyridyl ligands. Overcoming the baseline reactivity of this class of catalyst toward hydrogen evolution in aqueous media presents a major challenge for focusing this photochemical reactivity into desired transformations of more complex organic substrates. In this work, a photochemically induced aqueous ketone reduction catalyzed by [Cp*Ir(bpy)H][Cl] is reported and benchmarked to the competing hydrogen evolution reaction (HER). A detailed mechanistic study is conducted of the reduction of cyclohexanone with sodium formate delivering the reducing equivalent. Examining the kinetic profile of the cyclohexanone reduction revealed an unexpected induction period, suggesting a more complex underlying reaction mechanism where product selectivity was not simply a function of direct competition from a common excited-state reactive intermediate. The reaction kinetics were analyzed by variable time normalization analysis (VTNA), and the catalyst resting state was interrogated through operando light-emitting diode (LED) illumination 1H NMR spectroscopy. These and other mechanistic studies provide insight into a photochemical reaction pathway to ketone reduction that is distinct from that previously reported for HER.
Citation
Photocatalytic Transfer Hydrogenation in Water: Insight into Mechanism and Catalyst Speciation
David M. Kaphan, Kelsey R. Brereton, Rachel C. Klet, Ryan J. Witzke, Alexander J. M. Miller, Karen L. Mulfort, Massimiliano Delferro, and David M. Tiede
Organometallics 2021 40 (10), 1482-1491
DOI: 10.1021/acs.organomet.1c00133