Controlling Amphiphilic Polymer Folding beyond the Primary Structure with Protein-Mimetic Di(Phenylalanine)
Abstract
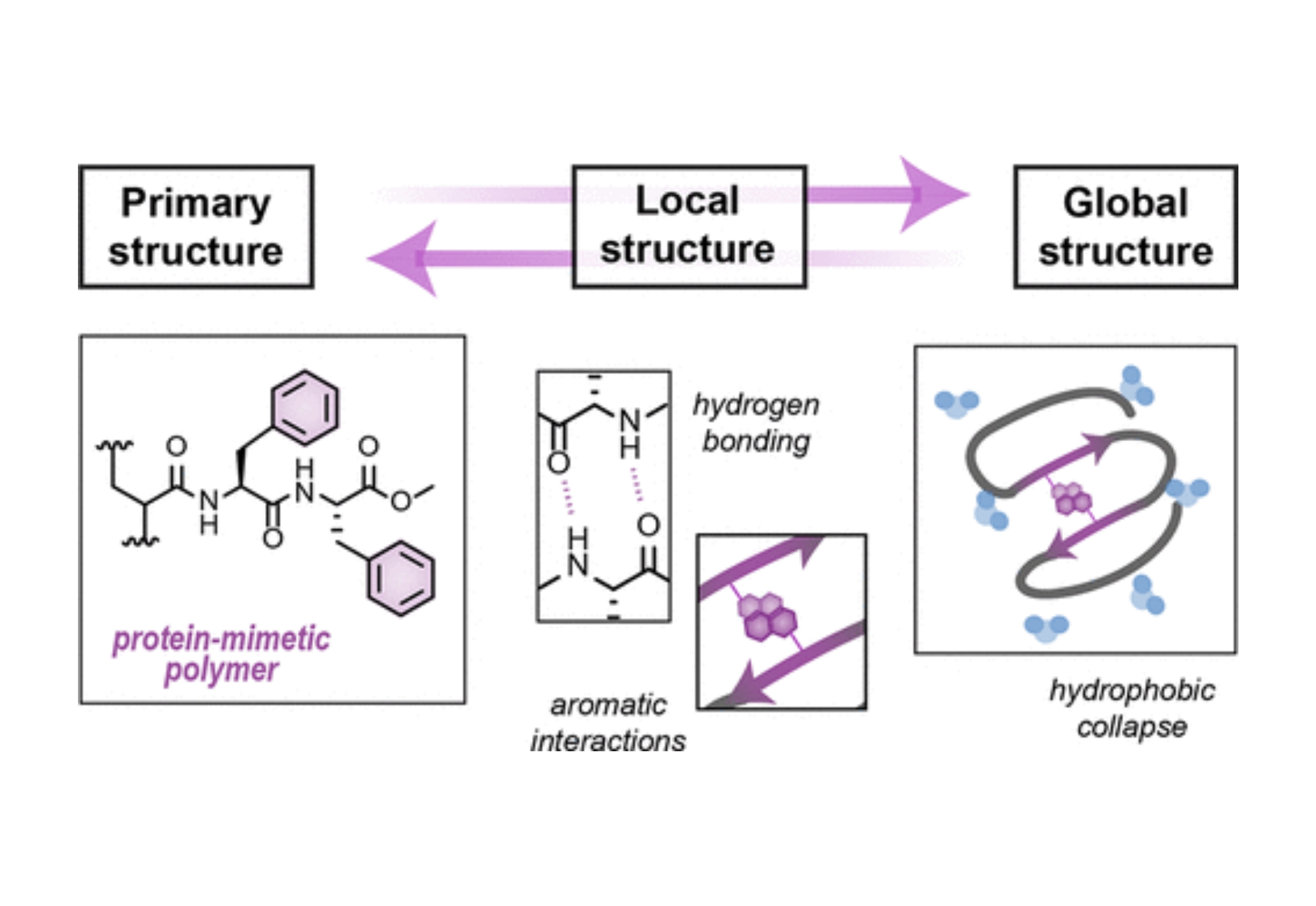
While methods for polymer synthesis have proliferated, their functionality pales in comparison to natural biopolymers–strategies are limited for building the intricate network of noncovalent interactions necessary to elicit complex, protein-like functions. Using a bioinspired di(phenylalanine) acrylamide (FF) monomer, we explored the impact of various noncovalent interactions in generating ordered assembled structures. Amphiphilic copolymers were synthesized that exhibit β-sheet-like local structure upon collapsing into single-chain assemblies in aqueous environments. Systematic analysis of a series of amphiphilic copolymers illustrated that the global collapse is primarily driven by hydrophobic forces. Hydrogen-bonding and aromatic interactions stabilize local structure, as β-sheet-like interactions were identified via circular dichroism and thioflavin T fluorescence. Similar analysis of phenylalanine (F) and alanine-phenylalanine acrylamide (AF) copolymers found that distancing the aromatic residue from the polymer backbone is sufficient to induce β-sheet-like local structure akin to the FF copolymers; however, the interactions between AF subunits are less stable than those formed by FF. Further, hydrogen-bond donating hydrophilic monomers disrupt internal structure formed by FF within collapsed assemblies. Collectively, these results illuminate design principles for the facile incorporation of multiple facets of protein-mimetic, higher-order structure within folded synthetic polymers.
Citation
Controlling Amphiphilic Polymer Folding beyond the Primary Structure with Protein-Mimetic Di(Phenylalanine)
Jacqueline L. Warren, Peter A. Dykeman-Bermingham, and Abigail S. Knight
Journal of the American Chemical Society 2021 143 (33), 13228-13234
DOI: 10.1021/jacs.1c05659