Stereospecific Nickel-Catalyzed Reductive Cross-Coupling of Alkyl Tosylate and Allyl Alcohol Electrophiles
Abstract
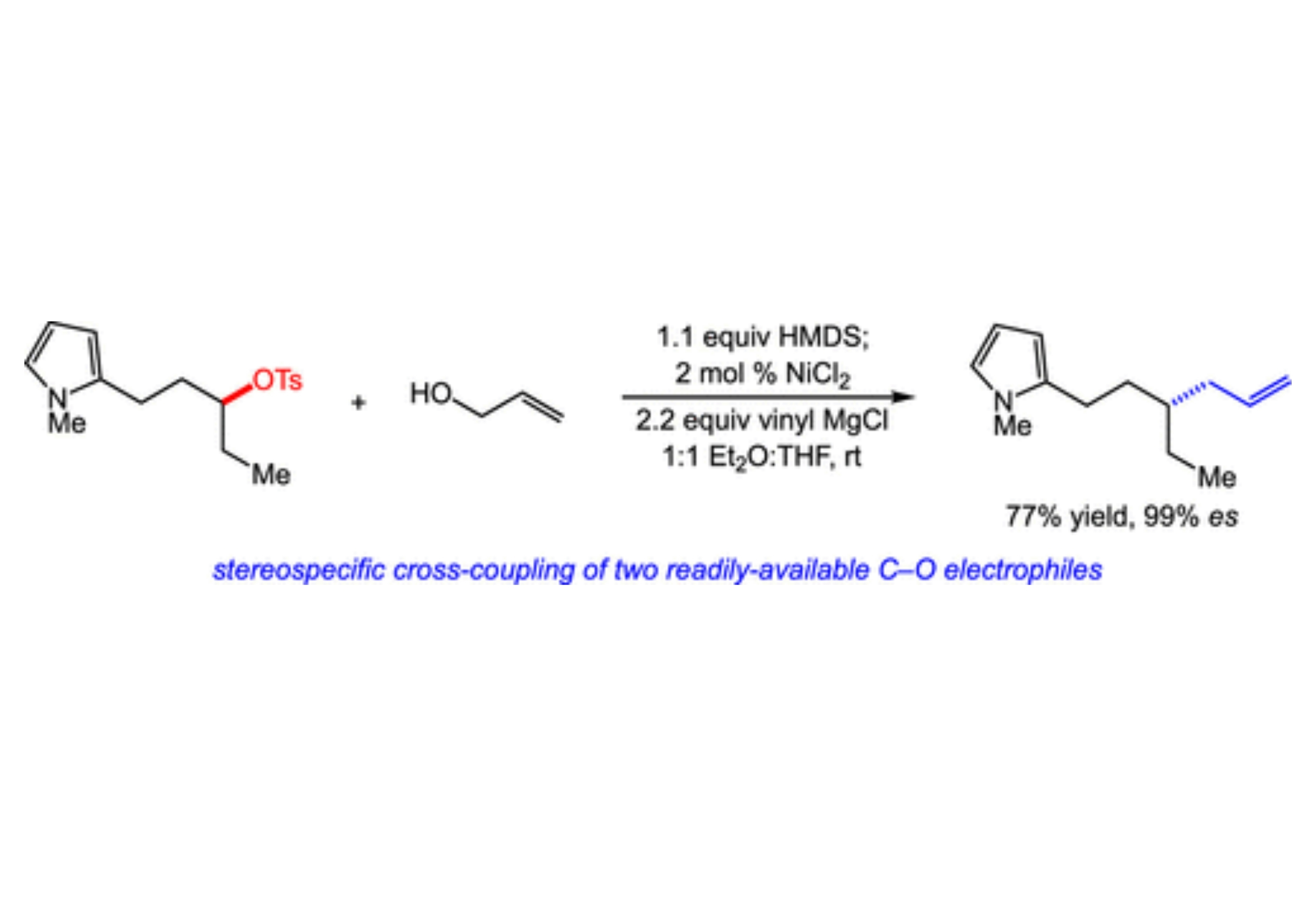
The stereospecific cross-coupling of easily accessed electrophiles holds significant promise in the construction of C–C bonds. Herein, we report a nickel-catalyzed reductive coupling of allyl alcohols with chiral, nonracemic alkyl tosylates. This cross-coupling delivers valuable allylation products with high levels of stereospecificity across a range of substrates. The catalytic system consists of a simple nickel salt in conjunction with a commercially available reductant and importantly represents a rare example of a cross-coupling involving the C–O bonds of two electrophiles.
Citation
Stereospecific Nickel-Catalyzed Reductive Cross-Coupling of Alkyl Tosylate and Allyl Alcohol Electrophiles
Quentin D. Tercenio and Erik J. Alexanian
Organic Letters 2021 23 (18), 7215-7219
DOI: 10.1021/acs.orglett.1c02616