Oligo(ethylene glycol) Side Chain Architecture Enables Alcohol-Processable Conjugated Polymers for Organic Solar Cells
Abstract
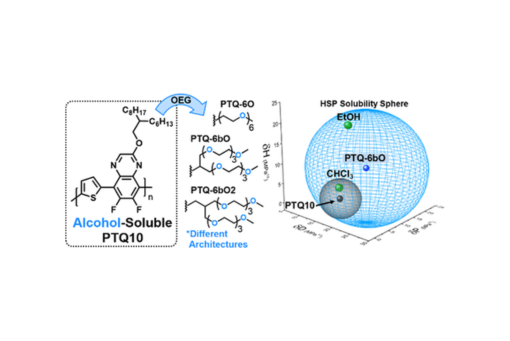
Achieving green-solvent solubility of conjugated polymers in truly green solvents such as alcohols has proven to be a significant challenge. In this work, we report the synthesis and characterization of three conjugated polymers derived from poly[(thiophene)-alt-(6,7-difluoro-2-(2-hexyldecyloxy)quinoxaline)] (PTQ10) with the goal of developing derivates which are more green-solvent-processable. The traditional alkyl side chains are replaced by various oligo(ethylene glycol) (OEG) side chains of different architectures, including one linear and two branched, all of which contain six ethylene glycol repeating units. It is determined that the linear OEG side chain architecture, even when sufficiently long, will not give desired green-solvent solubility shown by a small solubility capacity (R0). However, branched OEG side chains significantly improve solubility as R0 was increased from 4.7 of PTQ10 to 11.9 of PTQ-6bO/6bO2. Although the solution states of the polymers were vastly different, the solid-state morphologies were more similar as all three OEG-based polymers retained a predominately face-on molecular orientation similar to PTQ10. It was demonstrated that PTQ-6O devices showed the most comparable power conversion efficiencies (PCEs) to PTQ10 in bulk heterojunction solar cells, while PTQ-6bO2 and PTQ-6bO showed poorer performances. With one extra carbon in the side chain, PTQ-6bO2 showed higher PCE than PTQ-6bO, attributed to the improved aggregation properties and solid-state morphology of PTQ-6bO2, highlighting the importance of OEG side chain architecture. This work serves to develop important guidelines for future alcohol-soluble materials for green-solvent-processed OPVs.
Citation
Oligo(ethylene glycol) Side Chain Architecture Enables Alcohol-Processable Conjugated Polymers for Organic Solar Cells
Justin Neu, Stephanie Samson, Kan Ding, Jeromy James Rech, Harald Ade, and Wei You
Macromolecules 2023 56 (5), 2092-2103
DOI: 10.1021/acs.macromol.2c02259