Pseudomonas Virulence Factor Pathway Synthesizes Autoinducers That Regulate the Secretome of a Pathogen
Abstract
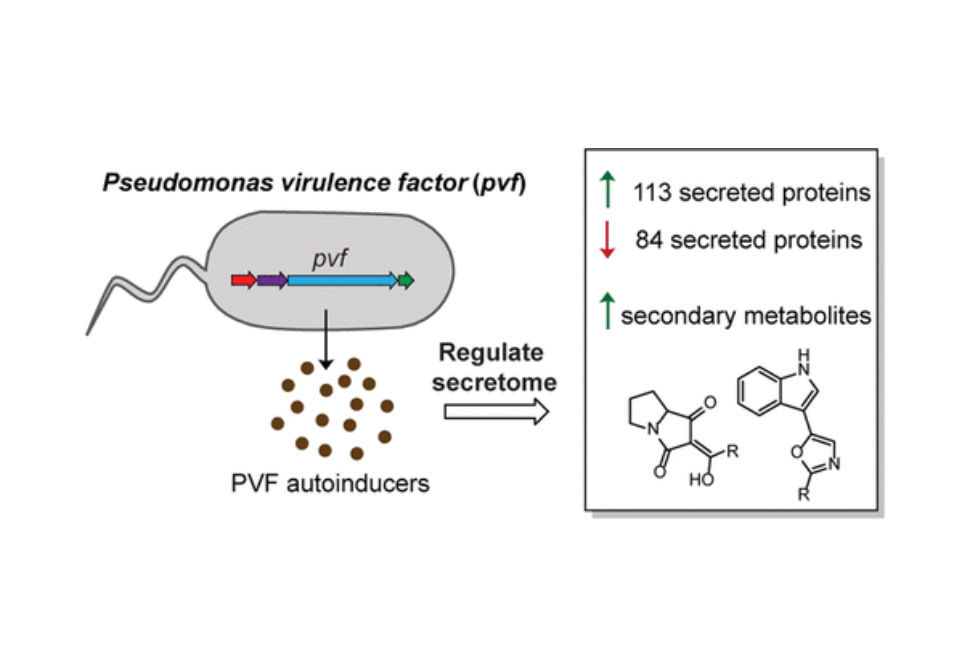
Cell-to-cell communication via chemical signals is an essential mechanism that pathogenic bacteria use to coordinate group behaviors and promote virulence. The Pseudomonas virulence factor (pvf) gene cluster is distributed in more than 500 strains of proteobacteria including both plant and human pathogens. The pvf cluster has been implicated in the production of signaling molecules important for virulence; however, the regulatory impact of these signaling molecules on virulence had not been elucidated. Using the insect pathogen Pseudomonas entomophila L48 as a model, we demonstrated that pvf-encoded biosynthetic enzymes produce PVF autoinducers that regulate the expression of pvf genes and a gene encoding the toxin monalysin via quorum sensing. In addition, PVF autoinducers regulate the expression of nearly 200 secreted and membrane proteins, including toxins, motility proteins, and components of the type VI secretion system, which play key roles in bacterial virulence, colonization, and competition with other microbes. Deletion of pvf also altered the secondary metabolome. Six major compounds upregulated by PVF autoinducers were isolated and structurally characterized, including three insecticidal 3-indolyl oxazoles, the labradorins, and three antimicrobial pyrrolizidine alkaloids, the pyreudiones. The signaling properties of PVF autoinducers and their wide-ranging regulatory effects indicate multifaceted roles of PVF in controlling cell physiology and promoting virulence. The broad genome distribution of pvf suggests that PVF-mediated signaling is relevant to many bacteria of agricultural and biomedical significance.
Citation
Pseudomonas Virulence Factor Pathway Synthesizes Autoinducers That Regulate the Secretome of a Pathogen
Ashley M. Kretsch, Gina L. Morgan, Katie A. Acken, Sarah A. Barr, and Bo Li
ACS Chemical Biology 2021 16 (3), 501-509
DOI: 10.1021/acschembio.0c00901